Ionic solids1/25/2024  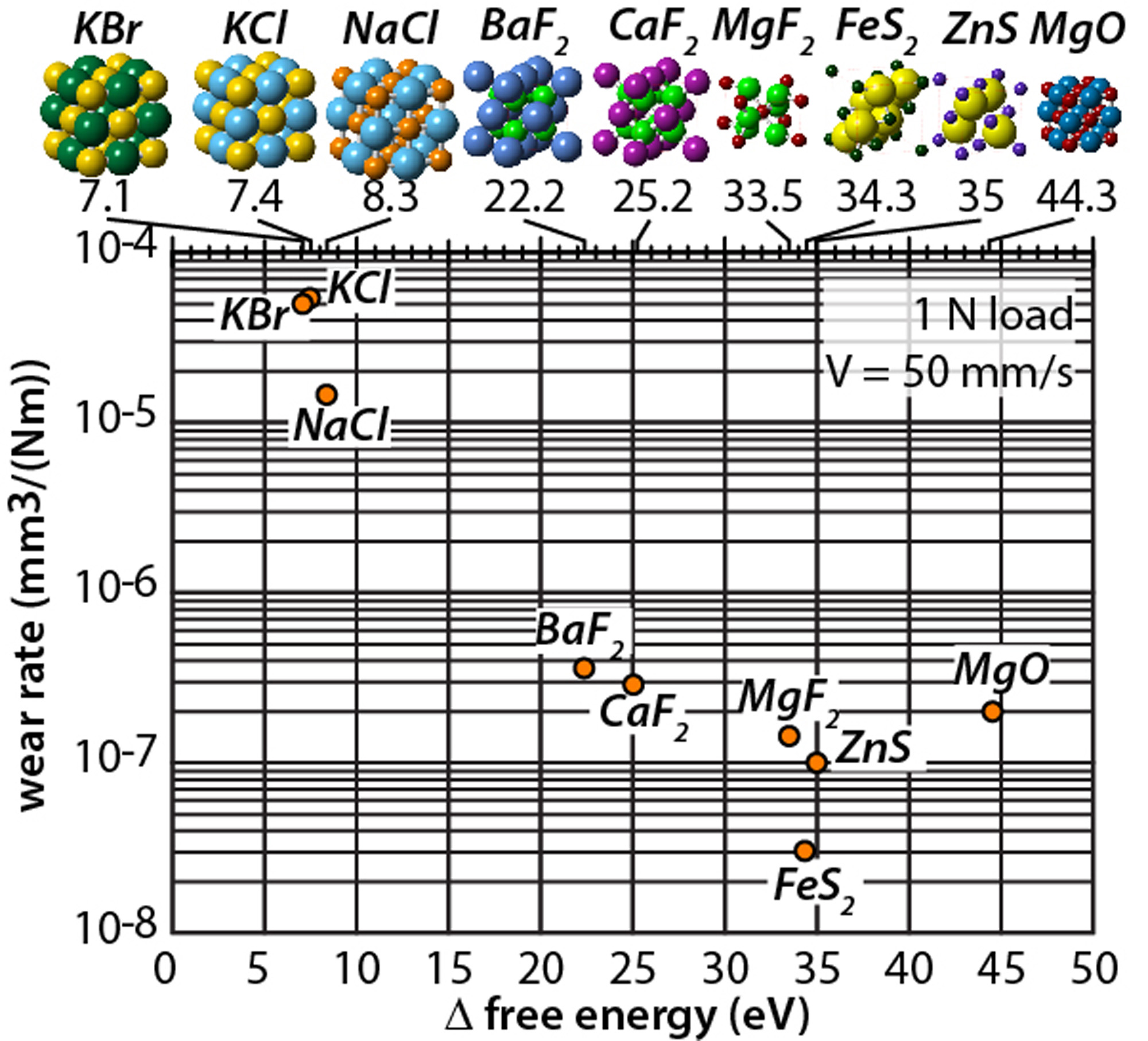 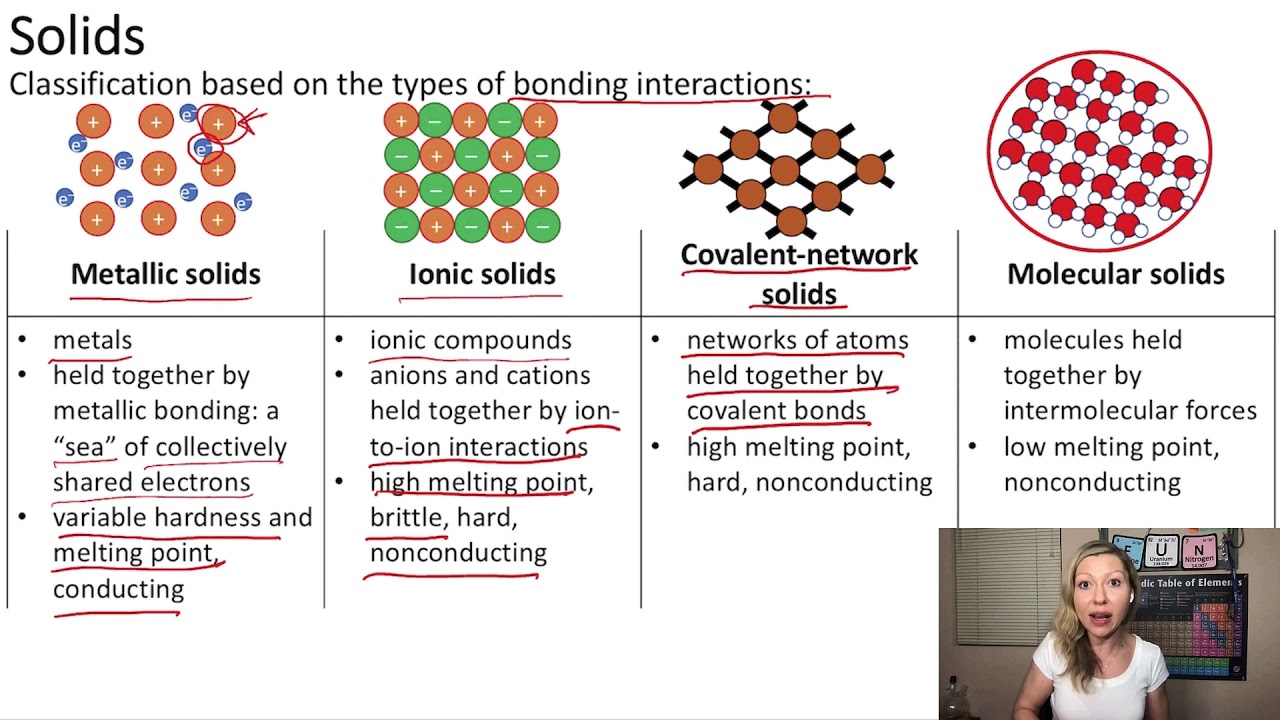 
There are four different types of crystalline solids, all with different properties: ionic, covalent network, metallic and molecular. Watch a video lesson on the types, properties and examples of ionic solids, and see how they differ from other types of solids like molecular solids and metallic solids. It also explains why cesium chloride has a different structure from sodium. Learn what ionic solids are, how they are composed of cations and anions held together by electrostatic forces, and why they tend to be hard, brittle and have high melting points. Ionic solids tend to have high melting points and are rather hard. This page explains the relationship between the arrangement of the ions in a typical ionic solid like sodium chloride and its physical properties - melting point, boiling point, brittleness, solubility and electrical behavior. Ionic solids consist of positively and negatively charged ions held together by electrostatic forces the strength of the bonding is reflected in the lattice energy. The enthalpies of fusion also increase smoothly within the series: benzene (9.95 kJ/mol) < naphthalene (19.1 kJ/mol) < anthracene (28.8 kJ/mol). Crystalline solids are the most common type of solids, whose structure consists of a regular, repeating pattern of atoms or molecules in three dimensions. The major types of solids are ionic, molecular, covalent, and metallic. For example, the melting points of benzene (C 6H 6), naphthalene (C 10H 8), and anthracene (C 14H 10), with one, two, and three fused aromatic rings, are 5.5☌, 80.2☌, and 215☌, respectively. While the intermolecular forces are strong enough to hold the molecules in place, molecular solids typically have lower melting and boiling points than metallic, ionic, or network atomic solids, which are held together by stronger bonds. See examples of ionic solids, such as sodium chloride, and how they adopt different 3-D crystalline patterns. Covalent molecules held together by intermolecular forces form molecular solids. Substances that consist of large molecules, or a mixture of molecules whose movements are more restricted, often form amorphous solids. Metals and ionic compounds typically form ordered, crystalline solids. For similar substances, the strength of the London dispersion forces increases smoothly with increasing molecular mass. Learn how to represent the structure of an ionic solid using a particulate model, a method that maximizes the attractive forces between opposite charges and minimizes the repulsive forces between like charges. Figure 10.37 The entities of a solid phase may be arranged in a regular, repeating pattern (crystalline solids) or randomly (amorphous). 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
